Lyme disease was first described in 1975 in 51 residents of Old Lyme, Lyme, and East Haddam CT. They were all diagnosed as having a unique form of multiple joint arthritis. In 1982, a PhD student, Willy Burdorfer, discovered the organism responsible for the disease. The organism was found within a deer tick (Ixodes scapularis) on Long Island, NY. This organism is described as a slender, spirally undulating bacteria, now known as a spirochete. In 1983, the spirochete was named after their original discoverer, Borrelia burdorferi.
In 1984, the first case of dog Lyme arthritis was reported. The same signs – multiple intermittent joint arthritis, fever, and flu-like symptoms were all observed. As more medical professionals became aware of this disease, more patients were diagnosed from suffering from the infection.
In 2000, a quote in the veterinary medical literature stated Lyme disease “might not be the most prevalent equine disease you face with your horses, but it does exist, and can seriously impact a horse’s health. Although documented cases of Lyme disease in horses are relatively rare, there are indications that the disease is on the increase.” In 2006, Lyme disease had now become the most common tick-borne disease in the US.
In 2003, research was being conducted at Cornell University on Lyme disease in horses. A research team led by Dr. Tom Divers reported 50% of horses in the Northeastern United States are positive for exposure to Lyme disease. This indicates at least 50% have been bitten by a tick carrying the Borelli burdoferi organism. Dr. Divers’ team also reported success using intravenous oxytetracycline as a treatment for horses infected with Lyme disease. In addition, his research showed some protection against the disease in ponies vaccinated with a recombinant DNA canine vaccine; although safety and frequency of injections has not been determined.
Ticks in Ixodes classification have 35 different species in North America and 250 species worldwide. The ticks are capable of transmitting viruses, bacteria, rickettsia, protozoans, and nematodes (Borrelia). These are each different types of organisms, some able to live on their own and others requiring other life forms to stay alive. They each cause different types of infection from different types of organisms.
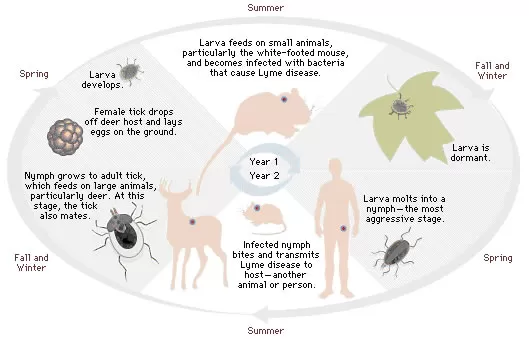
The deer tick (Ixodes scapularis) is also commonly called a black legged tick. It has a requirement for three different hosts to complete its life cycle from egg to adult. Each feeding stage requires one vertebrate blood meal for its development. The adult female lays the EGG which then becomes a LARVA. The Larval stage now must complete a blood meal to become the next stage, a NYMPH. The nymphal stage must also complete a blood meal before changing into an ADULT. Each of the three feeding stages (Larval, Nymph, Adult ticks) is capable of causing infection. As the tick grows in size with each life stage it will feed on larger hosts. Humans and horses are likely hosts for adult ticks or nymphal stage ticks. Horses are more likely to have a higher prevalence of Lyme disease than humans because ticks stay on horses longer. Research has shown that ticks must be attached for at least 24 hours to transmit their disease. The tick holds the organism in its simplified digestive system, known as a hindgut. A tick attaches to a host with its very sharp mouthparts. It then salivates and regurgitates into the host an anti-clotting agent so it can get a ready supply of host blood. While the tick is attached to the host it uses its mouth parts to suck and ingest blood of the host. It then sends this blood to its hindgut where it mixes with the organism. The tick then regurgitates back into the host as it continues to feed. If the tick had the Borrelia organism in its hindgut, it has now been deposited in the host. Not only does the host become infected, but is now serves as reservoir of Borrelia for the next tick to ingest when it attaches and continues to spread the disease.

The adult stage of the deer tick is active fall to spring when the temperature is above 40 degrees. Adults feed on large mammals (horses, dogs, deer and human). Deer are reservoirs for adult ticks, but not for the organism itself. This means the deer do not have the disease in a high population, but they do act as a great host for ticks and can now spread ticks throughout a wide area with their movements. After engorging, the female tick drops off in a place to lay her eggs and the life cycle continues. Larvae will metamorphasize into nymphs, now the size of a poppy seed. The 8-legged nymphs are most active in May, June, July, August. Larval activity is seen in May from fall eggs. The second and much larger larval activity peaks in August from the more successful spring breeding season. If infected as a larval stage; the infection is now transmitted to the nymphal stage. The nymph now seeks a host for a blood meal. This host is normally a mouse or smaller mammal, or bird. As it is the size of a poppy seed, it is easily undetected. But the tick now has the second opportunity to get infected from an infected host; get blood meal, drop off and turn into sexually active mature blacklegged ticks.
As stated previously, an infected nymph passes on to an adult; now the 3rd carrier. Typically this is the only stage we find; if found and removed before 24 hours transmission can be prevented. The nymphal stage has the greatest Lyme disease transmission as the season is the longest with the highest number of hosts available (May – June).
Earlier we explained the spirochete is transmitted to host via salivation and/or regurgitation. The spirochete is primarily found in the hind gut of tick, but it can be in salivary glands of the tick as well. Studies have shown it takes a minimum of 24 hours of tick attachment for spirochete transmission to the host. Due to the fact we can now see this adult life stage, and it takes some time to pass on its disease, we can interrupt transmission of the disease before it occurs. In addition as adults are most active fall to spring when temperatures are over 40 degrees, activity is often decreased in winter climates. Additionally, horses and humans are less active and have less exposure time as the weather cools down. Therefore, disease transmission decreases decidedly during adult season, even though up to 50% of adult ticks may be infected as we often remove them and the weather patterns are in our favor.
Making the diagnosis of Lyme disease can be challenging. The disease mimics many other diseases.
Fever, muscle aches, fatigue = viruses
Joint pain = arthritis
Neurologic signs (weakness and incoordination) = mimic many other diseases
Infected horses often have vague and variable signs. We see multiple different signs, sometimes more than one at a time. For example, signs most often reported include:
- stiffness/lameness
- muscle tenderness
- hyperaesthesia (increased or altered sensitivity to sensory stimuli)
- resentment of touch or pressure
- swollen joints (rare)
- behavioral changes
- Fever and edema are unlikely (more likely a result of Anaplasma phagocytophilia infection, or a dual infection)
- Unwillingness to work, lethargic, “grumpy”
We often have to rule out joint problems (osteoarthritis), muscle soreness or injuries (rhabdomyolysis), neurologic issues (EPM), and swollen joint issues (OCD fragments). The diagnosis is difficult due to fact the most common presenting complaint is lameness, which may be intermittent and non specific. It may take a few days to weeks to develop clinical signs. There is not a specific type of lameness and there are numerous causes for lameness complaints. The lameness may be episodic or recurring and chronic. It can be the forelimbs, the pelvis, the spine, or the rear limbs. Central nervous system problems (encephalitis) have also been described. Eye problems described as similar to moon blindness (recurrent uveitis) have also been described. On the bright side: heart, liver, kidney problems have not been reported in horses, as they have in dogs and humans, and Lyme disease is rarely fatal in any species.
Just as in human, the ideal diagnosis is by testing the tick itself. This is rarely done, as quite often owners report the tick bite occurred weeks or months previously, or that a tick has never been seen. Therefore, we use a variety of blood tests.
Before going on, a few words of vocabulary may help to understand the discussion:
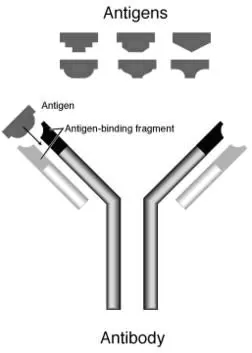
Antibody – protein normally present in the body or produced in response to an antigen. The neutralizing of antigens by antibodies is called the immune response.
Antigen – (bacteria) introduced into the body by the tick as it feeds. When they are detected, the immune system begins to create antibodies to fight the intruders.
Antigen binding fragment – at tip of antibody which identifies and binds (neutralizes) antigens
IFA or ELISA Test (“titer”): This test is performed by applying the patient’s serum to an antigen-coated slide and adding an anti-body conjugate. The slide and conjugate are made by different companies (several different manufacturers), which affects the test dramatically. Different labs use different dilutional schemes for their titers. Interpretation of the test is technician dependent and read as a number of dilutions. A higher dilution indicates more antibodies. Unfortunately, titers do not equate to clinical signs or degree. Titers don’t equate into degree of infection. This test measures the amount of IgG an animal has against Lyme disease. If the test is reported as a high titer it can be interpreted as an exposed animal, an animal who has mounted a significant immune response, an infected and sick animal, or a recently cleared infection. If the test is reported as low, this can be interpreted as not infected, a cleared infection, or an animal that has not yet mounted an immune response. Therefore, this test lacks specificity and has many false positives (animals testing positive who do NOT have the disease).
Other tests commonly utilized include:
- Western blot (4 of 4 bands) – the “gold standard” takes several days (4) for the test to be completed. One must also transport samples to reference lab, and the results again can be ambiguous and requires repeating, especially within the first few weeks after infection. This test is also quite expensive.
- Whole cell diagnostic methods (Kela units) measure the IgG antibodies produced to numerous antigens on the “whole” spirochete. The persistence of the whole cell antigens produce antibodies that are present and remain elevated even when spirochetes are reduced or eliminated.
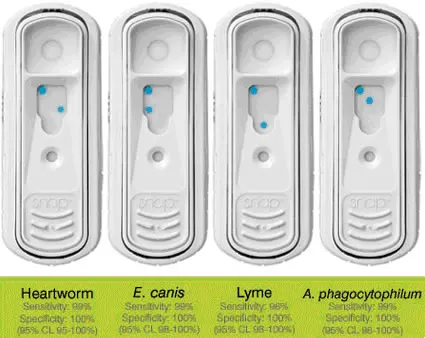
- SNAP ELISA test = C6 antibody. Introduced in 2004 as an in house dog test. The C6 antibody is associated with the variable region on the antibody which is only expressed when in the presence of live spirochetes. This test declines rapidly and significantly after effective treatment has removed the organism, as the previously mentioned variable region would no longer be expressed. This test will detect antibodies against Lyme disease as early as 3 weeks post infection and has shown to be 100% consistent with Western Blot results.
- Lyme Multiplex test – this blood test looks at 3 different Outer Surface Proteins (OSP); A, C, and F. By measuring the levels of the three proteins, some conclusions may be made on recent or long standing infection, versus exposure without infection. This test has become available this year (2011) and also takes a few weeks to show a recent infection.
The test takes 8 minutes and uses 3 drops of blood. It can be done stall side. The Lyme disease SNAP test is comparable or superior to the traditional 2-tiered testing of IFA and Western Blot. The fact that a decrease in the amount of antibodies against C6 can be seen quickly will indicate a successful therapeutic outcome for Lyme patients. A drop of 50% or greater indicates a successful treatment. In 2005, a study validated this test in horses. A study reported in 2006 showed this test to have a high specificity and sensitivity (rare false positives or false negatives). This is a quick, reliable, in-house test, has been validated for horses.
- A study measured 164 horse samples – 106 positive with Western blot, 109 positive with SNAP (all 3 tested positive with IFA) = 99.4% sensitivity (it did not miss any positives, but identified a few more which may or may not be true positives)
- A repeated test 60 – 90 days after completion of antibiotics of therapy can be used to assess success of treatment and status of the horse.
Notes on the SNAP test: Any color development indicates a positive result. You should not draw a correlation b/w the color intensity of the sample spot and the level of infection. It takes 8 minutes to read final result; waiting longer does not increase the accuracy of the test.
As this is a bacterial infection, antibiotics are the basis of treatment. As reported in 2005, 100% of infected horses in one study were completely cleared of infection by using intravenous oxytetracycline. The study also compared two other antibiotics, doxycycline and ceftiofur (Naxcel); only to find that both were only 50% effective at removing the infection. We also know that doxycycline is poorly absorbed by the horse, and extended treatments may be necessary in order to utilize the horse’s immune system to rid the disease while the doxycycline keeps the infection in check. In the study at Cornell, re-infection was common in both the doxycycline group and Naxcel group, but not the oxytetracycline group.
Body soreness, muscle soreness, and/or joint pain can be controlled with non steroidal anti-inflammatory agents (NSAIDs). Often treatment is only necessary for 3 to 5 days while the antibiotic is mounting its attack against the organism. In addition, the joints that are inflamed are at risk for developing chronic problems or cartilage issues from the inflammatory mediators within the joints. Therefore, cartilage protective agents (chondroprotection) are often used simultaneously for the best outcome in many cases.
Acupuncture has also shown to be helpful in cases where attitude and body soreness are the major complaints.
As the injected antibiotic chelates (or grabs) circulating calcium, it must be diluted and given slowly to ensure the heart and skeletal muscle do not have any problems. A typical treatment takes 3- 5 minutes. Also, it is not a treatment for untrained professionals as the antibiotic can be irritating to veins if not administered so that the entire dose is completely within the vein.Our treatment utilized intravenous oxytetracycline daily for 5 to 10 days and then uses a form of oral oxytetracycline for 2 additional weeks. Most horses (and their veins) can accommodate this treatment protocol without the use of intravenous catheters. Our success rate for achieving negative test results after treating is over 80%. Reasons for the less than perfect test results are threefold: re-infection (as horses return to their areas of infection); incomplete treatment (owners unable to or horses unwilling to complete the oral treatment); or incomplete resolution, but a greater than 50% reduction in circulating C6 levels (as the SNAP test only records color –although it can vary from faint to dark blue).
This protocol takes into consideration that intravenous oxytetracycline ONCE daily is superior to oral doxycycline or intramuscular Naxcel twice daily. This is due to higher tissue concentrations of oxytetracycline compared to low levels of oral doxycycline. Those that did respond to doxy or Naxcel in the Cornell study showed a significant decline in antibody level during treatment but also showed antibody levels increased after treatment was discontinued in 75% of each treatment group.
- These two drugs appear to inhibit reproduction of, but not eradication of B. burgdorferi.
- Doxycycline also has potent anti-inflammatory effects which may allow horses to show improvement in their lameness signs regardless of the drug’s effect on the organism.
Many different methods of prevention have been attempted and utilized. The recombinant DNA canine vaccine did show it was effective in preventing Lyme disease in ponies challenged with infected ticks 3 weeks after a three-dose regimen. The vaccine appears to inhibit the spirochete within the tick. This means as the tick ingests the blood meal from the host which is laced with the antibodies from the vaccine it will inhibit or kill the spirochete in the tick’s hindgut. Therefore, ticks known to be carrying the spirochete do not infect the host when placed on them. Unfortunately, we do not how frequently to vaccinate and also do not know if it is safe to give to a horse which may have been previously infected or is currently not known to be infected. There are no safety studies. At this time we cannot recommend using a dog Lyme vaccine on your horse.
What we do know is that dogs bring ticks into homes and fields and our friends live in close proximity to people and horses. Dogs roll in leaves and run into tick habitats where the larval, nymphal, and adult stages lay waiting for their next blood meal. Dogs are reported to be 50% more likely to get Lyme disease than humans and are now used by human doctors to determine if Lyme disease is in the area. If a family member complains of symptoms similar to Lyme disease, quite often treatment is initiated if the family pet has also been diagnosed with the disease. Therefore, protecting pets often may all that be necessary to protect you and your horse. (Note: cats can get it too)
Horse prevention: Examine your horse on a daily basis; remove nymphs and adults immediately. Remember it takes at least 24 hours of attachment to transmit the disease. Be sure to check the neck, base of the mane, under the tail and the ears. If you see a tick, remove it by grabbing it as close to the skin as possible with fine tweezers. Pull straight up slow and steady. You can then put the tick in a sealed container (no alcohol) for testing. Once the tick is removed apply alcohol or antibiotic ointment to the site.
Try to keep clear of tick-infested area. If not possible, use insect repellent with permethrins in spring, summer and fall– although none are approved for use on horses. As horses sweat, just like us humans, whatever we spray on them topically with dilute and ultimately become inactive. There are now permethrin wipes made for dog use that can be used for horses. We recommend using them before venturing into tick infected areas by wiping each leg, the main and tail once an hour while in the area.
Clean up leaf/brush piles in our yard, your paddocks, and on your farm. These are great areas for the white-footed mouse to live and continue to cycle of Lyme disease on your property. Mow your fields to again minimize the number of mice and also to minimize the vegetation ticks like to climb on. It is also recommended to use wood chips or gravel between lawns and wooded areas to restrict tick migration into your grass paddocks or yards.
Stack wood in dry areas where ticks are less likely to enjoy a safe haven. As we stated earlier, deer serve to spread the ticks wherever they go. Do not feed deer on your property and, if you need to, plant deer resistant plants to inhibit them from approaching too close.
You can also purchase or construct bait boxes to treat wild rodents with an acaracide (kills ticks). These are available for home use in some feed supply stores or you can make them by placing treated cotton balls inside old paper towel or toilet paper rolls. Treat the cotton balls with permethrins in order to have the rodents take the cotton balls back to their homes where they will make them part of the bedding and continuously rub up against them removing any ticks. This can help reduce ticks by more than 50%.
By protecting your pets to help to protect yourself and your horse.
The future looks to remove the source of infection, that is, the organism inside the white-footed mouse. A project was undertaken to vaccinate the mice to assess its effectiveness. 900 white footed mice in 12 different forested sites in CT were trapped and vaccinated with dog vaccine. The rodent’s antibodies kill the bacterium inside the tick – preventing the tick from spreading the disease to the next host. After vaccinating 55% of the mouse population in targeted areas, researchers measured an overall reduction in the prevalence of Lyme disease transmission in nymph-stage ticks. As trapping and vaccinating mice is a never ending and labor intensive (and expensive) undertaking, the emphasis is now on the need to develop an oral vaccine (like Rabies). Initially, a large amount of money for funding the research to develop the vaccine is required. Then there will need to be trials conducted that measure the effectiveness in numerous different areas before a product will be available for use. This will take YEARS to develop.
There is no evidence of transmission directly from animal to human, and one cannot get infected from the urine of an infected animal. A horse also cannot get infected from eating a tick or portion of a mouse (in baled hay). The spirochete is not a highly resistant organism in the environment; it would probably be killed by the acidity in the stomach and small intestine and by the digestive enzymes. Plus, it must be introduced into the skin or directly into the bloodstream.
SUMMARY of LYME DISEASE IN HORSES
CLINICAL SIGNS
- Vague and Variable:
- Stiffness, lameness, muscle tenderness,
- Hyper sensitivity, swollen or sore joints,
- Behavioral changes – lethargic or “grumpy”
TESTING
- 4DX SNAP TEST = C6 Antibody (at least 3 weeks post infection)
- INCIDENCE = 25 – 30 %
TREATMENT
- IV OXYTETRACYCLINE once daily for 5-10 days followed by oral Oxytetracycline powder for 2-3 weeks (> 80% success rate)
RETESTING
- At least 58 days after treatment has been completed
PREVENTION
- Canine Vaccine – no safety data or dosing schedule available yet
- Tick repellents on dogs
- Remove ticks from horses within 24 hours
- Use permethrin sprays, avoid tick areas
- Control mouse population – clean up brush, mow fields, bait boxes
- Do not encourage deer